Thomson Atomic Model
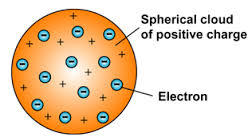
The "plum pudding" model is one of several scientific models of the atom. First proposed by J. J. Thomson in 1904 soon after the discovery of the electron, but before the discovery of the atomic nucleus, the model represented an attempt to consolidate the known properties of atoms at the time: the electrons are negatively-charged particles and the atoms are neutrally-charged.
In this model, atoms were known to consist of negatively charged electrons. Though Thomson called them "corpuscles", they were more commonly called "electrons". At the time, atoms were known to be neutrally charged. To account for this, Thomson knew atoms must also have a source of positive charge to balance the negative charge of the electrons. He considered three plausible models that would satisfy the known properties of atoms at the time: 1) Each negatively-charged electron was paired with a positively-charged particle that followed it everywhere within the atom. 2) Negatively-charged electrons orbited a central region of positive charge having the same magnitude as all the electrons. 3) The negative electrons occupied a region of space that itself was a uniform positive charge (often considered as a kind of "soup" or "cloud" of positive charge).
Thomson chose the third possibility as the most likely structure of atoms. Thomson published his proposed model in the March 1904 edition of the Philosophical Magazine, the leading British science journal of the day. In Thomson's view:
[...] the atoms of the elements consist of a number of negatively electrified corpuscles enclosed in a sphere of uniform positive electrification, [...]
The colloquial nickname "plum pudding" was soon attributed to Thomson's model as the distribution of electrons within its positively-charged region of space reminded many scientists of "plums" in the common English dessert, plum pudding.
Thomson original article
REFERENCES
Encyclopædia Britannica. Available in: https://www.britannica.com/science/Thomson-atomic-model. Access in: 24/08/2018.
SEARS; ZEMANSKY; YOUNG; FREEDMAN. Física IV. Ótica e Física Moderna. 12ª Ed. Pearson, São Paulo. p. 185-189.




















0 comments
Sign in or create a free account